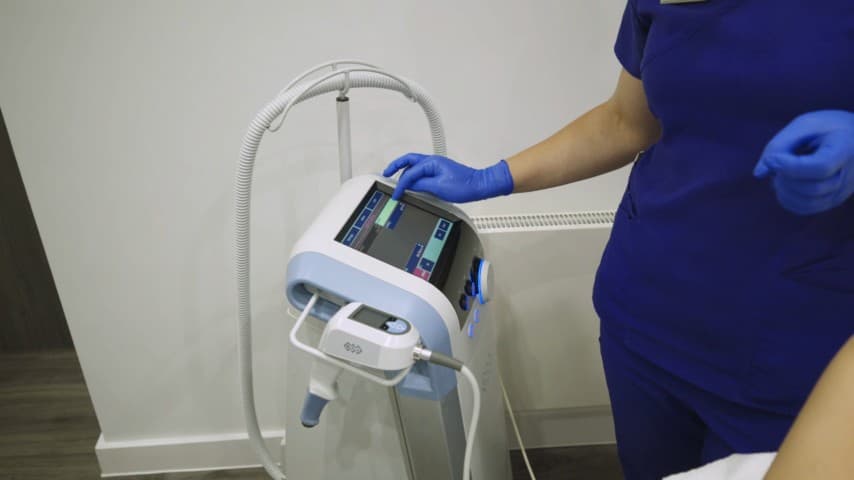
Ultra Femme 360 intimate rejuvenation in Maidenhead
Ultra Femme 360 is a non-surgical radiofrequency treatment using BTL technology for intimate tissue rejuvenation — addressing concerns such as laxity, reduced sensation, dryness and mild stress urinary leakage. At Berkshire Aesthetics in Maidenhead, suitability is assessed clinically and treatment is delivered with sensitivity and discretion within our doctor-led, CQC-registered clinic. Suitability, expected outcomes and maintenance are confirmed during your doctor-led consultation in Maidenhead.
Book ConsultationClinically reviewed by
Dr Selena Langdon — Medical Director, MBBS
Last reviewed:
Information is general and not a substitute for a medical consultation. Suitability is confirmed during assessment.
Why we start with a consultation
Every treatment begins with a medical assessment. We need to understand your medical history, current health, skin condition and concerns before prescribing any procedure. This protects your safety and ensures the treatment is clinically appropriate for you.
During your consultation, your clinician will explain what the treatment can realistically achieve, any risks specific to you, and whether an alternative might be more appropriate. Consultations are carried out by our doctors or advanced aesthetic practitioners, depending on the treatment or concern being assessed.
All bookings are subject to our Booking Conditions.
Not sure what you need?
Try our free AI skin analysis to identify your main concerns and see suggested treatments.
Start free skin analysis →Doctor-led approach
- Treatment suitability is assessed by our doctors or advanced aesthetic practitioners during a clinical consultation, depending on the treatment
- Prescribing responsibility sits with Dr Selena Langdon (GMC 6159259)
- No same-day injectable treatments for new patients
- Read our full Consultation & Safety Standards
Clinical overview
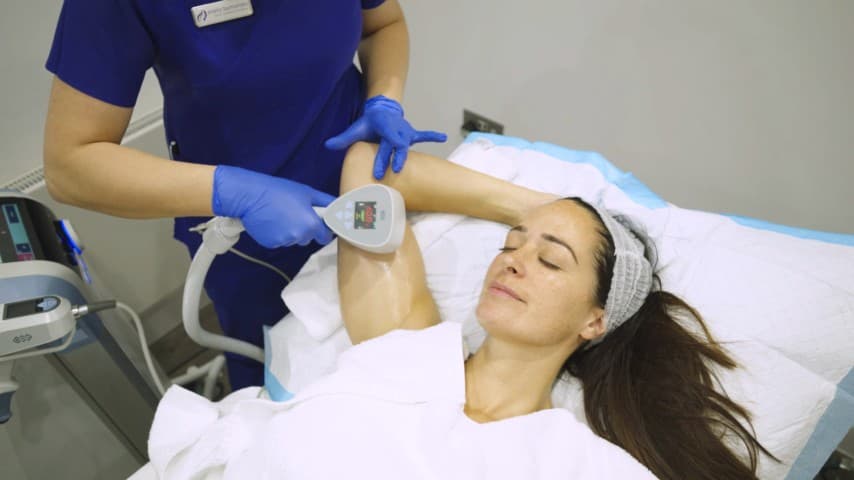
Ultra Femme 360 uses radiofrequency energy delivered via both internal and external applicators to gently heat intimate tissues, stimulating collagen production and tissue remodelling. The BTL device is designed specifically for non-surgical intimate rejuvenation. The treatment can address a range of concerns including tissue laxity, reduced sensation, vaginal dryness, mild stress urinary leakage and cosmetic appearance of the external intimate area. It is non-invasive, requires no anaesthesia and involves no downtime. We use Ultra Femme 360 for patients who want non-surgical improvement in intimate tissue tone and function. A typical programme involves 3 sessions spaced 1–2 weeks apart, with maintenance treatments recommended every 6–12 months. Suitability is confirmed during a sensitive and thorough consultation where we assess your concerns, medical history, expectations and whether this treatment — or an alternative — is the most appropriate option.
Treatment Areas
Internal Vaginal
Ultra Femme 360 delivers radiofrequency energy internally to stimulate collagen and elastin production in the vaginal walls. Treatment addresses vaginal laxity, dryness and reduced sensation by promoting tissue remodelling and tightening.
External Vulvar
The external applicator treats the vulvar area with radiofrequency energy to improve skin laxity, texture and appearance. Treatment stimulates collagen production for gradual tightening and rejuvenation of the external tissues.
Combined Internal and External
A comprehensive treatment session addresses both internal vaginal laxity and external skin quality in a single appointment. The combined approach provides more complete results for patients experiencing multiple intimate wellness concerns.
Key Benefits
These are the main clinical and practical reasons patients choose Ultra Femme 360 intimate rejuvenation in Maidenhead. Your treatment plan is tailored after consultation rather than selected from a fixed package.
- Non-surgical intimate rejuvenation with no downtime
- Internal and external applicators for comprehensive treatment
- Addresses tissue laxity, reduced sensation, dryness and mild stress urinary leakage
- Quick, comfortable sessions — each treatment takes approximately 8–15 minutes
- Stimulates collagen production for ongoing tissue improvement
Suitable For
Suitability depends on your goals, skin or tissue characteristics, medical history, and tolerance for downtime. These criteria are reviewed in detail during your assessment.
- Intimate tissue laxity following childbirth or age-related changes
- Reduced sensation or dryness affecting intimate comfort and confidence
- Mild stress urinary leakage that is not caused by significant anatomical issues
- Patients who want non-surgical improvement with no downtime
- Those seeking to improve the cosmetic appearance of the external intimate area
Not Suitable For
Some patients should avoid or defer treatment for safety reasons. We screen carefully before any procedure and explain alternatives where relevant.
- Pregnancy or planning to become pregnant in the near term
- Active infection or inflammation in the treatment area
- Significant pelvic organ prolapse (requires specialist assessment)
- Implanted electronic devices in the pelvic area
- Patients with unrealistic expectations of what non-surgical treatment can achieve
Final suitability is confirmed during a medical consultation and assessment.
When we don't recommend it
A consultation may identify scenarios where a different treatment route is safer or more effective for your outcome goals.
- If there is significant pelvic organ prolapse — this requires specialist gynaecological assessment and radiofrequency treatment alone is unlikely to be sufficient.
- If there is active infection or inflammation — treatment must be delayed until the area is fully settled.
- If urinary leakage is moderate to severe — we may recommend EMSella or Core to Floor as part of a broader plan, or refer for specialist assessment.
- If expectations are for surgical-level tightening — we will explain the realistic scope of non-surgical radiofrequency treatment and discuss alternatives where appropriate.
- If another treatment or combination would provide a safer or more effective outcome — for example, combining with EMSella for pelvic floor strengthening.
What Happens During Treatment
We follow a consultation-first process with clear consent and aftercare. You can also review our full consultation standards.
- 1Sensitive and private clinical consultation including your concerns, medical history and goals
- 2Discussion of expected outcomes, realistic timelines, limitations, risks and alternatives
- 3The procedure uses both internal and external applicators to deliver radiofrequency energy to the targeted tissues
- 4Treatment is comfortable — most patients describe a gentle warming sensation with no pain
- 5Each session is brief (approximately 8–15 minutes) and you can return to normal activities immediately
Downtime & Aftercare
Recovery guidance helps protect your result and reduce avoidable complications. We provide written aftercare and safety-net advice at every appointment.
- No downtime — resume normal daily activities immediately after treatment
- Avoid sexual intercourse for 48 hours following each session
- Avoid hot tubs, saunas and swimming for 48 hours
- Mild warmth in the treated area may be felt but typically settles quickly
- If you notice unexpected or persistent symptoms, contact the clinic for advice
Results Timeline
Most treatments improve in stages rather than immediately. Timelines vary by treatment type, treatment area, and individual response.
- A programme of 3 sessions spaced 1–2 weeks apart is typically recommended.
- Some patients notice improvement after the first session, with ongoing improvement as collagen remodelling progresses.
- Maintenance sessions are recommended every 6–12 months to sustain results.
How Long Results May Last
- •Results are maintained with regular maintenance sessions, typically every 6–12 months.
- •Longevity varies by individual, tissue condition and lifestyle factors.
- •Ongoing collagen stimulation means results can continue to improve between sessions.
Individual results vary. View our results and photography standards
Risks & Side Effects
Every medical treatment carries potential risks. We discuss these in plain language before treatment so you can make an informed decision.
Common, Usually Temporary
- •Mild warmth or tingling during and immediately after treatment (settles quickly)
- •Slight redness in the external treatment area (temporary)
- •Temporary increase in sensitivity in the treated area
Rare but Serious
- •Prolonged discomfort or swelling (uncommon; contact the clinic if concerned)
- •Burns or blistering (very rare with controlled radiofrequency delivery)
- •Any unexpected or worsening symptoms should be assessed promptly
When to Contact Us Urgently
- •Persistent discomfort, swelling or unusual discharge following treatment
- •Any signs of infection such as increasing redness, heat or tenderness
- •Any symptoms that concern you or are different from what was discussed
Frequently Asked Questions
These answers cover common practical questions. If you need individual advice, please book a medical consultation.
Quick Facts
Treatment Time
Approx. 8–15 minutes per session
Downtime
None — avoid intercourse and hot tubs for 48 hours
Results Visible
Improvement often noticed after first session; ongoing over course
Sessions
3 sessions, 1–2 weeks apart; maintenance every 6–12 months
Delivered by
Dr Selena Langdon — Medical DirectorMBBS, CoolSculpting Complications Expert, Aesthetic Doctor of the Year, JCCP Ambassador
Why Berkshire Aesthetics
- Doctor-led clinic — all treatments clinically overseen by Dr Selena Langdon (MBBS)
- CQC registered — view our registration
- Consultation-first approach — we assess suitability before recommending treatment
- Clear complaints process — we take patient feedback seriously
This content is for informational purposes only and does not constitute medical advice. Individual suitability for any treatment is assessed during a consultation. Always seek the guidance of a qualified medical professional with any questions regarding a medical condition or treatment.
- CQC registered clinic — regulated and inspected
- Doctor-led clinical oversight by Dr Selena Langdon (GMC 6159259)
- Complaints process and patient rights
Page last reviewed: 2026-02-10
Reviewed by Dr Selena Langdon, Medical Director (GMC 6159259) — Last reviewed: 2026-02-10
Plan your next step with pricing, consultation guidance, and related treatment pathways.
Compare Treatment Options
Use these clinical comparison pages to understand differences in indications, longevity and downtime.
Alternatives You May Want to Consider
Every patient is different. These treatments address similar concerns and may be more suitable depending on your goals and medical history.

EMSella
Non-invasive pelvic floor strengthening using HIFEM technology. Fully clothed, no downtime.
- Pelvic floor strength
- Non-invasive
- No downtime
Exilis Ultra
Non-invasive skin tightening and body contouring using combined radiofrequency and ultrasound energy.
- Skin tightening
- Body contouring
- No downtime
Core to Floor
Combined BTL programme using Emsculpt Neo for core strengthening and EMSella for pelvic floor rehabilitation.
- Core strengthening
- Pelvic floor rehab
- Non-invasive
Further Reading & Clinical References
The following external resources provide independent clinical and regulatory information.
- Radiofrequency skin treatments— DermNet
Ready to Start Your Journey?
Book a consultation with our expert team today.
